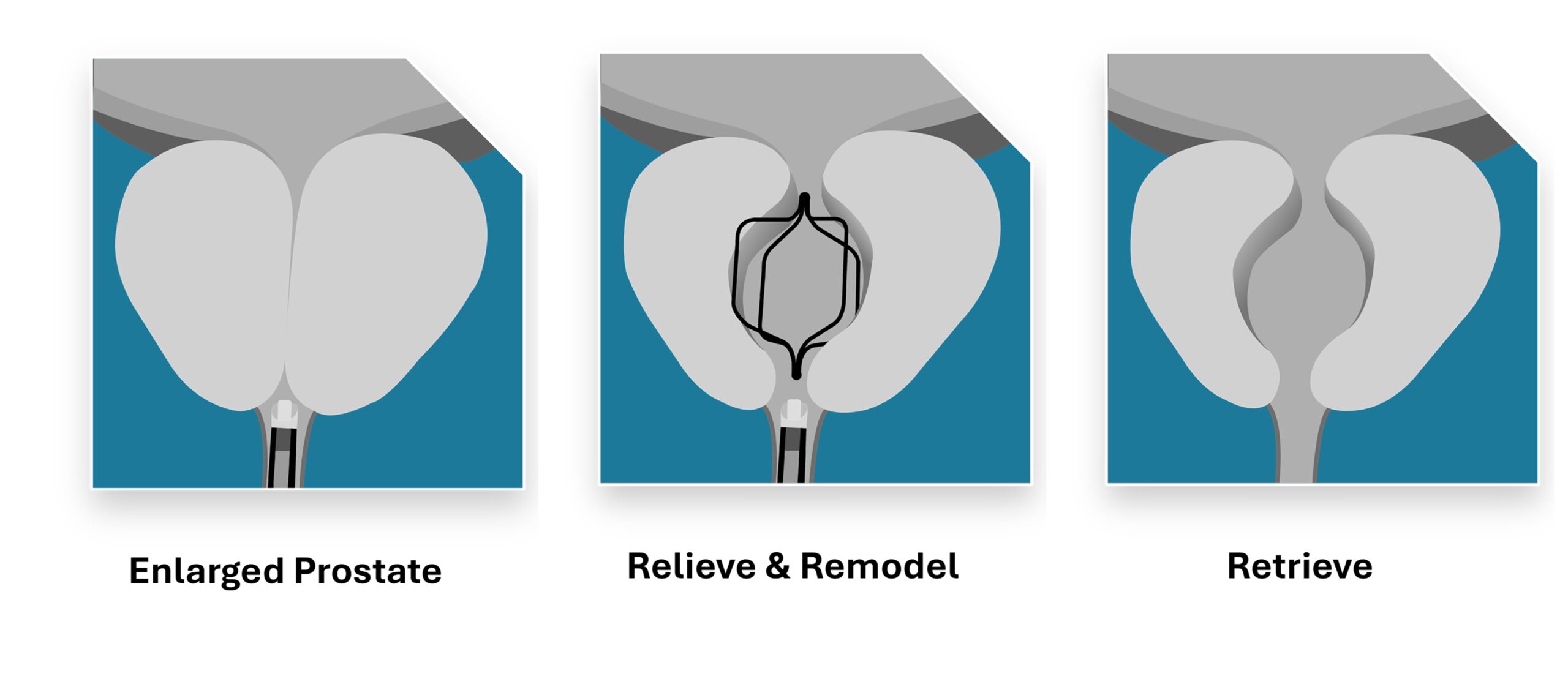
Prodeon Medical has achieved FDA 510(k) clearance for its novel Urocross® Expander System, a non-permanent implant aimed at addressing urinary symptoms related to benign prostatic hyperplasia (BPH). This innovative device is designed to remodel obstructed prostatic tissue through a minimally invasive procedure. Unlike traditional treatments, the Urocross procedure does not require the permanent placement of implants, which allows for future diagnostic and therapeutic options to remain intact.
The Urocross® Expander System utilizes readily available flexible cystoscopes to place the implant, which can be retrieved within six months. Clinical data supporting the FDA clearance demonstrated significant improvements in urinary symptoms, quality of life, and urinary flow after the retrieval process. Importantly, these improvements occurred without negatively impacting sexual function, underscoring the system’s potential to enhance overall patient care.
Clinical Trials and Data Supporting Clearance
The FDA’s clearance of the Urocross® Expander System was backed by a robust array of clinical trial data. This included findings from the Expander-1 Feasibility Trial and the Expander-2 Pivotal Trial, a large-scale, multi-center randomized controlled trial involving 240 patients across 23 sites in the United States and Canada. The Expander-2 trial, which employed a 2:1 randomization ratio, provided critical insights into the effectiveness and safety of the Urocross system.
Key outcomes from the Expander-2 trial highlighted sustained improvements in urinary symptoms and overall quality of life for patients undergoing the Urocross procedure. This data is pivotal for establishing the system’s intended role in managing lower urinary tract symptoms (LUTS) associated with BPH.
About Prodeon Medical and the Urocross® Expander System
Prodeon Medical, based in the United States, specializes in developing cutting-edge, minimally invasive medical devices for urologic diseases. The company’s mission centers on innovating therapies that challenge traditional treatment approaches while enhancing patients’ quality of life.
The Urocross® Expander System is engineered to remodel the obstructed prostatic urethra, utilizing a non-permanent, intentionally retrievable implant. The device is designed to remain in place for up to six months before being removed, ensuring that no permanent foreign material is left behind. This feature is especially significant for patients who may require additional treatments in the future.
Understanding Benign Prostatic Hyperplasia (BPH)
Benign prostatic hyperplasia is a prevalent condition affecting approximately 40 million men in the United States, with its incidence increasing significantly as men age. Symptoms associated with BPH can deteriorate over time, adversely affecting quality of life, sleep, and bladder health. As a result, there is a growing demand for minimally invasive treatments that not only provide effective relief but also preserve future therapeutic options for patients.
For further inquiries, media representatives can contact Tim Buckley, Executive Vice President of Prodeon Medical, via email at [email protected].