A recent study from the University of Barcelona has revealed a crucial molecular mechanism involved in how cells communicate through extracellular vesicles (EVs). These small particles have significant therapeutic potential, and the findings were published on November 28, 2025, in the Journal of Extracellular Vesicles. The research, led by Professor Albert Lu and María Yáñez-Mó, along with Carles Enrich, uncovers the role of the Commander protein complex in directing the entry and internal destination of vesicles within cells.
Understanding how cells send and receive signals is fundamental to developing new therapies and diagnostic tools. Professor Lu emphasized the importance of grasping how receptor cells process EVs, stating, “This knowledge is key to harnessing the therapeutic and diagnostic potential of these vesicles, as their effectiveness depends on being able to direct them and have them captured by the appropriate target cells.”
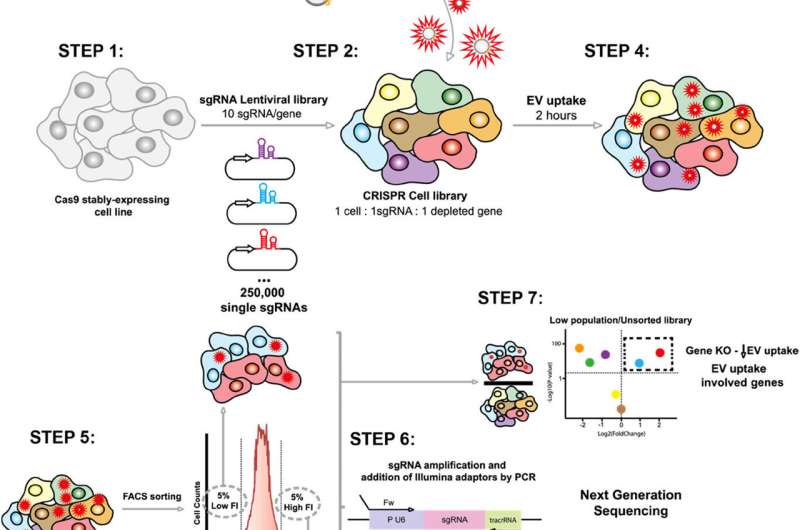
Innovative Methodology Utilizes CRISPR Technology
Extracellular vesicles act as biological messengers, transporting proteins, lipids, and nucleic acids between cells. The researchers employed an innovative approach using the CRISPR-Cas9 technology to explore the molecular mechanisms that govern EV uptake and internalization. By systematically deactivating each of the more than 20,000 human genes, they were able to assess the role of these genes in the process.
Cells were genetically modified to disable specific genes, then exposed to EVs labeled with a fluorescent dye. Using flow cytometry, the researchers measured how many vesicles each cell captured. Fluorescence-activated cell sorting (FACS) further allowed separation of cells based on their uptake capacity. This systematic and unbiased methodology enabled researchers to identify new regulatory mechanisms without relying on prior hypotheses.
The study found that the Commander endosomal recycling complex, composed of various proteins, serves as a fundamental regulator of vesicle uptake. The findings suggest that this mechanism is conserved across different human cell lines, indicating its potential universality, although its activity may vary depending on the cell type or physiological context.
Therapeutic Implications of Extracellular Vesicles
The implications of this research extend into therapeutic realms, as the ability of EVs to traverse membranes and target specific tissues makes them promising candidates for drug delivery systems. Professor Lu noted, “Understanding how their entry, intracellular trafficking, and delivery of their molecular cargo are regulated opens the door to designing EVs with controlled directionality, improving their efficacy in regenerative, oncological, or anti-inflammatory therapies.”
Ongoing research aims to delve deeper into the role of the Commander complex in controlling EV uptake and their intracellular fate. Researchers also plan to investigate whether alterations in this complex could impact cell communication in diseases such as cancer or neurodegenerative disorders. Ultimately, the goal is to manipulate this pathway to enhance communication between cells, optimizing the use of EVs as therapeutic and diagnostic tools.
For more information, refer to the original study: Miguel Palma‐Cobo et al, “Genome‐Wide CRISPR/Cas9 Screening Identifies the COMMANDER Recycling Complex as a Key Player in EV Uptake,” published in the Journal of Extracellular Vesicles.